What is halide?
A halide is a dual-phase in which one part is a halogen atom and the other part is an element or radical that is less electronegative or more electropositive than that of halogen to make a fluoride, chloride, bromide, iodide, astatide or hypothetically tennesside compound. Under suitable conditions, the alkali metals combine directly with halogens forming halides by using this general formula
MX (X= F, Cl, Br or I)
All metals present in Group 1 form halides that are white solids at room temperature.
Halide test
We will discuss halide ions (F, Cl, Br, I) tests by using silver nitrate and ammonia.
By Using Silver Nitrate Solution
We need a solution of halide ions. The step is adding a dilute nitric acid to acidify the solution. The nitric acid starts reacting with and removes the other ions present that might form precipitates with silver nitrate.
The second step is to add silver nitrate solution and the following products will be identified by the halide
| Ions present | Observation |
| F– | No precipitate |
| Cl– | White precipitate |
| Br– | A very pale cream precipitate |
| I– | A very pale yellow precipitate |
The formed precipitates of chloride, bromide and iodide precipitates are shown in the photograph below

The precipitates of chloride are identified easily but the rest of the two are quite similar to each other. They can be differentiated only in a side-by-side comparison. If the precipitates are exposed to light, all of them will change their colors i.e. grey or purple tints. The fluoride ions will not show any precipitates and the absence of a precipitate ion is unhelpful unless it is well known that a halogen is present otherwise, it will show that the chloride, bromide, or iodide is absent.
The chemistry of the test
The precipitates that are formed are insoluble silver halides: silver chloride, silver bromide or silver iodide. The following equation explains the formation
Ag+(aq) + Cl–(aq) → AgCl(s)
Ag+(aq) + Br– (aq) → AgBr(s)
Ag+(aq) + I–(aq) → AgI(s)
Silver fluoride is soluble so no precipitate is formed.
Ag+ (aq) + F– (aq) → Ag+(aq) + F– (aq)
Using ammonia solution to confirm the precipitate
A solution of ammonia is added to the precipitates
| Original Precipitate | Observation |
| AgCl | Precipitate dissolve to give a colorless solution |
| AgBr | Precipitate formed remain nearly unchanged by using dilute ammonia solution, while it dissolves in concentrated ammonia solution to give a colorless solution |
| AgI | Precipitate formed is insoluble in ammonia solution of any concentration |
Explanation
There are no insoluble ionic compounds. In water, if the concentrations of the ions in solution exceed a certain value, a precipitate will be formed which will be different for every different compound. This value can be considered as a solubility product. The expression for the solubility product of silver halides is given as
Ksp = [Ag+] [X–]
The square brackets express the molar concentrations having units of mol/L.
Conditions of forming precipitates
- There will be no precipitate formation if the actual concentration of the ions in a solution will produce less value than that of solubility product.
- There will be a precipitate formation if the product of the concentrations would exceed the value of the solubility product.
Necessarily the product of the ionic concentrations is never greater than the solubility product vale. Every time enough solid is precipitated to lower the ionic product to the value of the solubility product. The table given below enlists the solubility products from silver chloride to silver iodide (a solubility product of silver fluoride can’t not be described as it is too soluble).
| Ksp (mol2dm-6) | |
| AgCl | 1.8 x 10-10 |
| AgBr | 7.7 x 10-13 |
| AgI | 8.3 x10-17 |
You will notice that all compounds are very insoluble but become even less soluble as you go down from the chloride to iodide.
The Role of Ammonia
The ammonia comes in contact with silver ions to give a complex ion known as the diamminesilver(I) ion, [Ag(NH3)2] +. This reaction is reversible but the complex is very stable and the equilibrium position lies proficiently to the right. The equation for this reaction is given below:
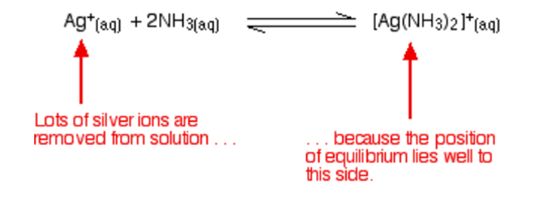
A solution that is in contact with one of the silver halide precipitates will contain a minute concentration of dissolved silver ions. The consequence of adding the ammonia is to lower this concentration still further. If the adjusted silver ion concentration is multiplied by the halide ion concentration is less than the solubility product, some of the precipitates will be dissolved to restore the equilibrium.
If the ammonia is concentrated this will take place with silver chloride and with silver bromide. The more concentrated ammonia pushes the equilibrium even further to the right that lowers the concentration of silver ions even more.
The silver iodide is very insoluble that the ammonia will not lower the silver ion concentration enough for the precipitate to dissolve.
An alternative Test using a concentrated Sulphuric Acid
By adding a concentrated Sulphuric acid to a solid sample of any one of the halide gives the results as follows
| Ion present | Observation |
| F– | Steamy acidic fumes (of HF) |
| Cl– | Steamy acidic fumes (of HCl) |
| Br– | Steamy acidic fumes (of Hbr) contaminated with brown bromine vapor |
| I– | Some HI fumes are seen with large amounts of purple iodine vapor and a red compound in the reaction vessel |
The only confusion that can occur between fluoride and chloride is that they behave identically under these conditions. They can be distinguished by dissolving the original solid in water and then testing with a silver nitrate solution. The chloride results in white precipitate while the fluoride produces none.
References
- https://www.chemguide.co.uk/inorganic/group7/testing.html
- https://chem.libretexts.org/Bookshelves/Inorganic_Chemistry/Supplemental_Modules_(Inorganic_Chemistry)/Descriptive_Chemistry/Elements_Organized_by_Block/2_p-Block_Elements/Group_17%3A_The_Halogens/1Group_17%3A_General_Reactions/Testing_for_Halide_Ions
- https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Supplemental_Modules_(Organic_Chemistry)/Alkyl_Halides/Reactivity_of_Alkyl_Halides/Reaction_of_Alkyl_Halides_with_Silver_Nitrate
- https://en.wikipedia.org/wiki/Halide
